
Cheryl Guttman Krader, BS, Pharm
Articles by Cheryl Guttman Krader, BS, Pharm

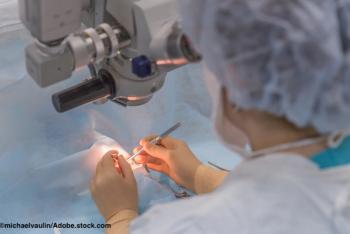
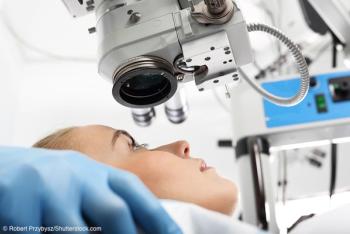

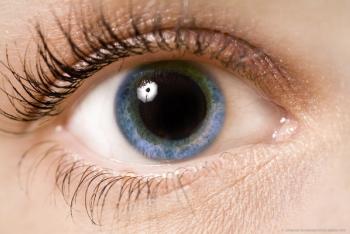

Results of the prospective TREX-DME study provide an evidence base for using a treat-and-extend protocol for administering anti-VEGF injections for eyes with diabetic macular edema.

Suprachoroidal triamcinolone acetonide (CLS-TA, Clearside Biomedical) injection resulted in visual and anatomic improvements in eyes with diabetic macular edema, particularly in those that were treatment-naïve. Multiple injections of the investigational treatment were well-tolerated and associated with a low incidence of IOP elevation.



The presence of vitreomacular adhesion (VMA) is associated with poorer, short-term anatomic, and functional outcomes in eyes with diabetic macular edema (DME) receiving anti-VEGF therapy, according to Márcio B. Nehemy, MD, PhD.

The efficacy of intravitreal bevacizumab (Avastin, Genentech) for improving vision in patients with center-involving diabetic macular edema (DME) has been demonstrated in several clinical trials.

All three anti-vascular endothelial growth factor (anti-VEGF) agents that are commercially available have demonstrated efficacy for the treatment of diabetic macular edema (DME). However, it is clear from the results of clinical trials that outcomes with bevacizumab are not as good as those obtained using ranibizumab or aflibercept, according to Jean-François Korobelnik, MD.

Analyses of data collected in the RISE/RIDE and VIVID/VISTA clinical trials provide important messages about the efficacy and safety of ranibizumab (Lucentis, Genentech) and aflibercept (Eylea, Regeneron) for treatment of diabetic macular edema (DME).

Abicipar pegol (Allergan/Molecular Partners) met its primary and key secondary endpoints and demonstrated an acceptable overall safety profile in a phase II trial investigating use of the novel anti-VEGF-A agent for treatment of diabetic macular edema (DME).

Sustained intraocular delivery of fluocinolone acetonide (FAc) using the FAc 0.19 mg intravitreal implant (Iluvien, Alimera Sciences) improves and slows progression of diabetic retinopathy (DR), according to findings of post-hoc analyses of data from the pivotal Fluocinolone Acetonide for Diabetic Macular Edema (FAME) trials.
Latest Updated Articles
 Topical squalamine offers novel, promising therapy for nAMD
Topical squalamine offers novel, promising therapy for nAMDDecember 11th 2017
 Insights from anti-VEGF pivotal DME trials
Insights from anti-VEGF pivotal DME trialsMay 10th 2017