Ophthalmology
Latest News
Video Series
Latest Videos
Podcasts
CME Content
More News

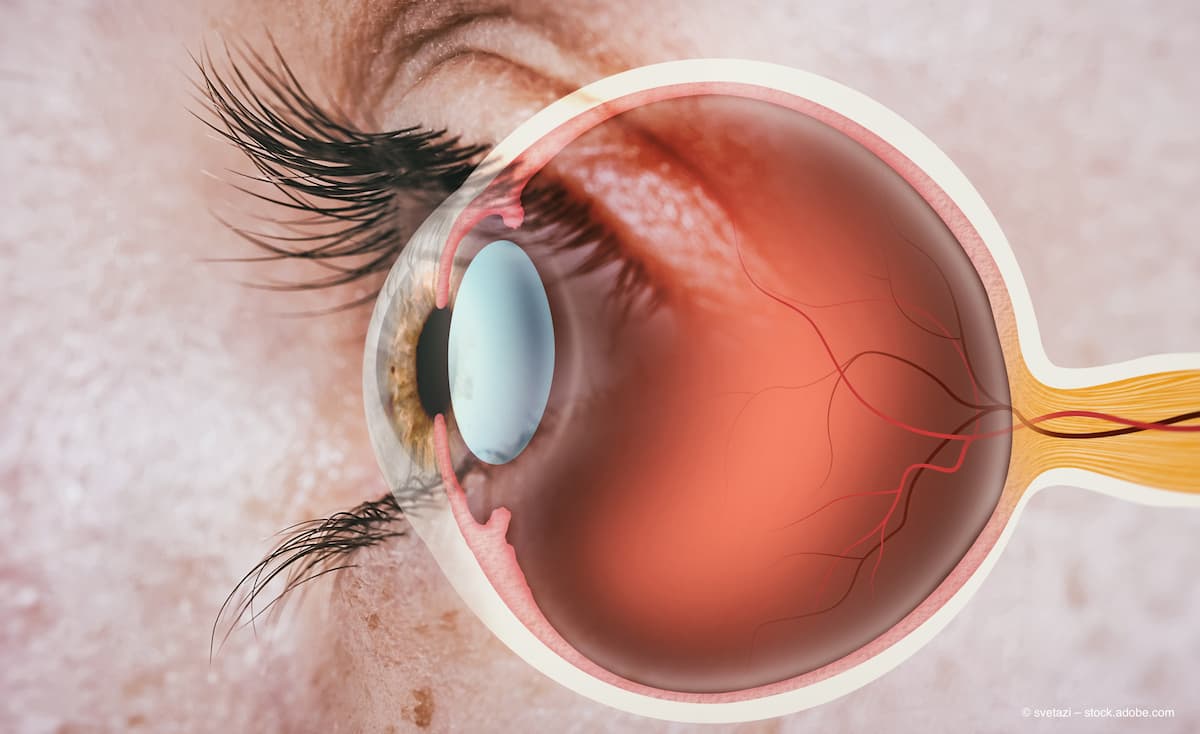
AI revolutionizes ophthalmology with enhanced diagnostics, personalized treatments, and improved surgical precision, offering better patient outcomes and efficient clinical trials.

Broadwood Partners, who is against the merger, holds approximately 27.5% of the outstanding common stock of STAAR, making them the largest outside stockholder of STAAR.

In addition to the proxy statement, STAAR has reached out to all stockholders to ask for their vote to adopt the merger agreement.
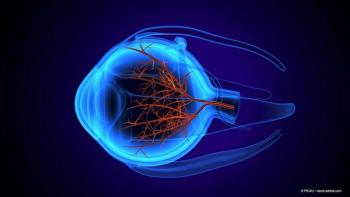
New research reveals significant inner retinal impairment in multiple sclerosis patients without optic neuritis, highlighting the need for further studies on macular health.


Research reveals a significant link between androgen exposure and central serous chorioretinopathy, aiding clinicians in assessing patient risk factors effectively.

Research reveals how neighborhood social factors impact patients with tractional retinal detachments, highlighting the need for improved access to care and support.


Matthew Starr, MD, discusses techniques for correcting astigmatism in patients lacking capsular support, enhancing visual outcomes surgery.

Lai spoke with Modern Retina about the study of PER-001, a novel therapeutic targeting the endothelin-1 receptor.

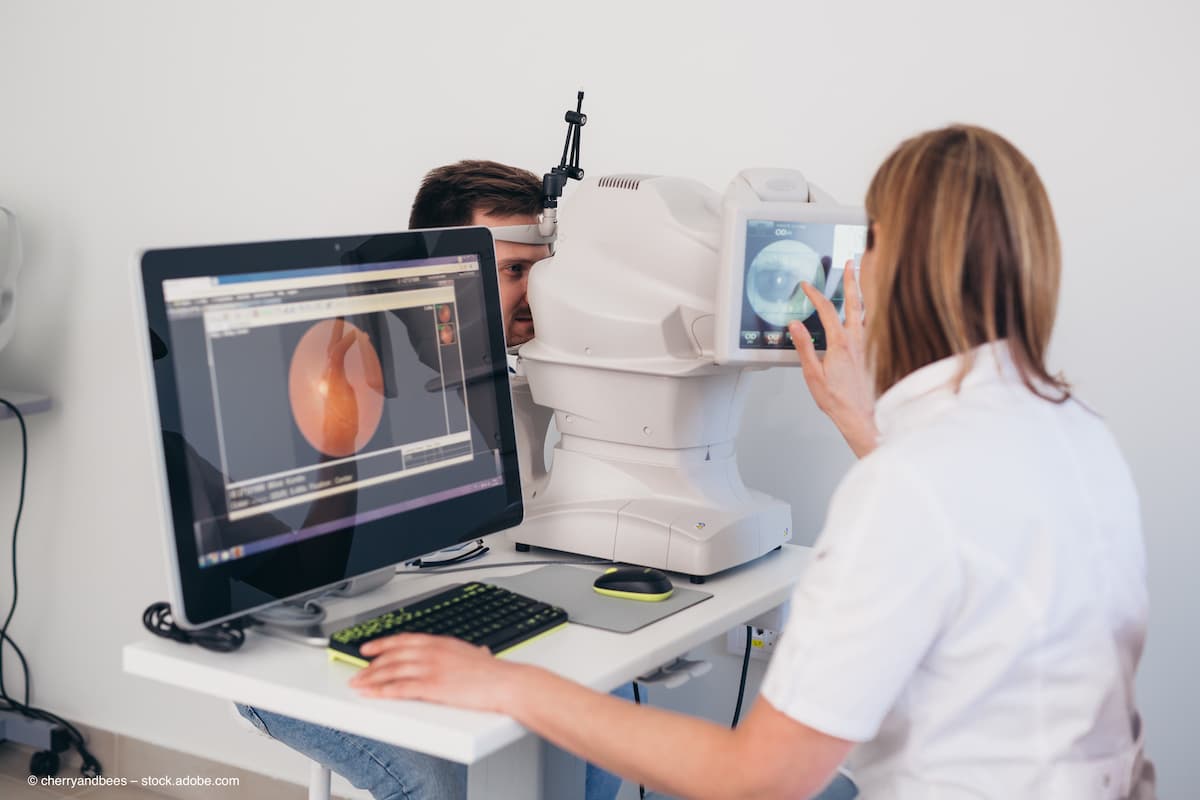
Avant Technologies and Ainnova Tech refine clinical trial protocol for Vision AI platform, aiming for FDA 510(k) approval to enhance diabetic retinopathy detection.
Clearside Biomedical secures Health Canada approval for Xipere, while exploring strategic options to enhance its suprachoroidal delivery platform and pipeline.

Andrew G. Lee, MD, shares essential insights on neuro-ophthalmology for retina specialists, emphasizing diagnosis techniques and collaboration with neuro-ops.

Re-Vana and Boehringer Ingelheim enter \$1B collaboration to advance sustained-release eye therapies
Re-Vana Therapeutics and Boehringer Ingelheim forge a $1 billion partnership to develop innovative extended-release therapies for eye diseases, enhancing patient care.
Mucormycosis emerges as a critical concern in diabetic patients with sudden vision loss, highlighting the need for prompt diagnosis and treatment.

New tariffs threaten the optical industry, prompting urgent reevaluation of sourcing strategies and pricing as deadlines approach for affected countries.

A recent study reveals cannabis use may lower the risk of proliferative vitreoretinopathy after retinal detachment repair, suggesting potential therapeutic benefits.

Patients express mixed feelings about AI in healthcare, favoring its use for documentation and administrative tasks while demanding transparency and safety standards.

Through the program, gene therapies are developed to treat patients with retinitis pigmentosa caused by pathogenic variants in the MERTK gene.

The report highlights clinical successes, innovation, research, and patient outcomes.

In a recent study, the researchers suggested that targeting the circuits responsible for visual acuity may be necessary in achieving optimal recovery of visual function and could aid in the development of vision restoration therapies.
Gore recently launched its silicone-free GORE IMPROJECT Plunger for 0.5-mL pre-filled syringes for the delivery of ophthalmic drugs.

Retinal vascular change or degeneration already has been associated with Alzheimer and Parkinson disease, and the findings suggest that neurologic diseases impact ocular health.

The drug has been reported previously to cause neuropathy and hypersensitivity reactions in breast cancer patients.

The cross sector initiative that intends to drive a shift from reactive to proactive health care by leveraging ocular data through the means of oculomics.