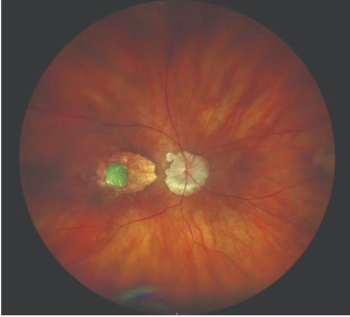
Geographic Atrophy
Latest News
Latest Videos

CME Content
More News
US eye medication shortages make up a sizable share of active drug shortages and last longer, driven by manufacturing issues, weak generics and unclear causes.

Steffen Schmitz-Valckenberg, MD, discusses how optical coherence tomography-based models may enable rapid, noninvasive assessment of functional loss in GA.

From artificial intelligence and IOL innovation to biosimilars, geographic atrophy, and postoperative eye protection, Joshua Mali, MD, FASRS, shares what he believes will define ophthalmology in the year ahead.

The results of a multicenter controlled clinical trial showed that the device resulted in “a significant improvement in visual acuity (VA) from baseline to month 12.

The deal will see MeiraGTx assume development rights to ZipBio’s experimental therapies targeting the complement pathway in geographic atrophy (GA).

Imaging and biomarkers are driving earlier, more individualized GA treatment
David S. Boyer, MD, outlines changes in GA evaluation, the role of complement inhibition, monitoring for neovascular conversion, and emerging therapies.

Nanoscope's new patent enhances its innovative MCO technology, promising vision restoration for retinal degeneration with strong safety results and market potential.

The Investigational New Drug (IND) application from Complement Therapeutics for CTx001 was previously approved by the FDA in October 2025.

Knowing what’s on the market for AMD and GA aids in preserving vision.
The novel system converts light into electrical signals to stimulate retinal cells.


This approach helps distinguish diseases associated with macular atrophy.

Atrophy Advisor aids physicians in personalizing treatment for geographic atrophy in age-related macular degeneration, enhancing patient care and outcomes.
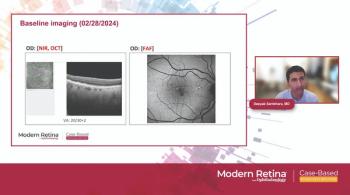
Lock in patient adherence with discussion of study results.
Results showed reduction in geographic atrophy progression and improved visual acuity.

Xelafaslatide is a small-molecule Fas inhibitor designed to protect key retinal cells, including photoreceptors, from cell death that occurs across multiple retinal diseases and conditions.

New research highlights iron dysregulation's role in dry AMD, suggesting transferrin as a promising treatment to slow disease progression.

K8 is a member of a new class of inflammasome-inhibiting drugs called kamuvudines.

Geographic atrophy management evolves with proactive imaging, personalized therapies, and real-world insights, enhancing patient care and treatment outcomes.

To help with education and awareness, Prevent Blindness is providing free, expert-approved educational resources on GA.

In the study, authors investigated the natural history of GA lesion incidence rates and analyzed potential risk factors for faster incidence of GA lesions.

New five-year data reveals SYFOVRE significantly delays geographic atrophy progression, enhancing understanding of age-related macular degeneration treatment.

A study reveals that lesion characteristics in geographic atrophy significantly influence growth rates, highlighting the need for targeted treatment strategies.
Retina specialists share their perspectives on evolving anti-VEGF therapy, pipeline treatments, and strategies to enhance patient outcomes.