Shedding light on the cause of retinal inflammation after gene therapy
The severity and duration of gene therapy–associated uveitis correlated with increasing vector dose.
The severity and duration of gene therapy–associated uveitis correlated with increasing vector dose. (Image Credit: Adobe Stock/julia_diak)
Reviewed by Kanmin Xue, MB-PhD, FRCOphth
A high vector dose of gene therapy can lead to the development of retinal inflammation after treatment, according to Kanmin Xue, MB-PhD, FRCOphth; Laurel Chandler, PhD; and Robert MacLaren, FMedSci, FRCOphth. The Oxford University researchers presented their observations during the Association for Research in Vision and Ophthalmology’s 2023 Annual Meeting, which was held in April.
Their results1 showed the potential for vision-threatening damage to the photoreceptors. Although retinal gene therapy demonstrates great promise for treating patients with a range of inherited retinal diseases, the prospect of developing inflammation may curtail its use and the success of the therapy, Xue said.
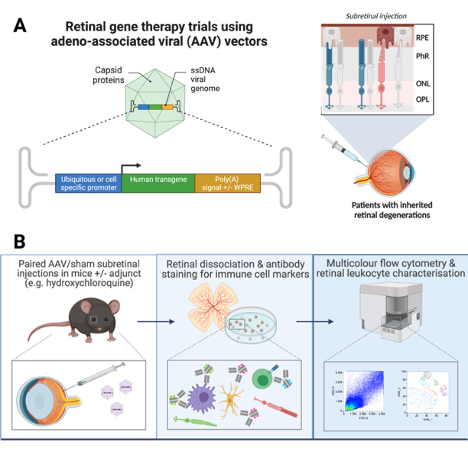
To gain an understanding of the inflammatory mechanism, the researchers reviewed clinical data from dose-escalating trials of adeno-associated viral (AAV) vector–mediated subretinal gene therapies. The observations of murine models that were treated with subretinal AAV vectors correlated with the clinical findings and were studied in depth using a range of molecular techniques, including analysis of retinal gene expression at early and late time points and characterization of immune cells by multiparameter flow cytometry (Figure).
Figure (A) Cases of retinal inflammation were encountered with dose escalation in multiple clinical trials of subretinal gene therapy using the AAV vector.
Figure (B) Clinical findings are correlated with observations in mouse models treated with subretinal AAV vectors expressing fluorescent marker, therapeutic transgene, or sham. At early and late time points (up to 6 weeks), retinal gene expression was analyzed by reverse transcription-quantitative polymerase chain reaction. Retinas were also dissociated for multiparameter flow cytometric analysis of immune cell populations. The effects of adjunctive hydroxychloroquine (19 μM) were assessed by coinjection with AAV vector.
Image courtesy of Kanmin Xue, MB-PhD, FRCOphth
Findings in patients and mice
The researchers reported that the results of multiple clinical trials showed that the severity and duration of gene therapy–associated uveitis correlated with increasing vector dose (typically ≥ 1011 genome particles per mL). The findings on optical coherence tomography suggested the breakdown of the blood-retinal barrier and immune cell infiltration of the retina.
The investigators cited a clinical trial2 in which chorioretinitis developed 2 weeks after high-dose (1 × 1011 gp) subretinal injection of an AAV2 vector expressing human REP1 to treat choroideremia. The retinal inflammation responded to systemic immunosuppression, but the treated eye showed a persistent reduction in visual acuity.
Another clinical trial3 showed subretinal infiltrates following dose-escalating (≥ 1 × 1011 vg) subretinal injections of an AAV8 vector expressing RPGR across cohorts of patients with X-linked retinitis pigmentosa. The retinal inflammation was associated with reduced retinal sensitivity that responded to escalating regimes of systemic with or without local immunosuppression.
In the murine models, according to the researchers, the subretinal AAV gene therapy activated pattern recognition receptors (such as toll-like receptors, which can detect foreign DNA) and resulted in release of proinflammatory cytokines (Tnf-α, Ifn-γ) by day 7,4 followed by retinal infiltration by monocytic phagocytes, CD8+/CD4+ T cells, and natural killer cells from week 2 onward. The latter is indicative of type 1 immunity typically seen against intracellular viruses.5 “We are searching for the antigen-presenting cells that initiate this immune response, with the microglia, retinal pigment epithelium (RPE), and Müller glia being potential suspects,” Xue commented.
Based on the finding of toll-like receptor 9 (TLR9) activation by AAV in the retina, the researchers administered the TLR9 inhibitor hydroxychloroquine as an adjunct during subretinal gene therapy. This improved AAV transduction by approximately 3-fold across a range of tissues, including the mouse retina in vivo, primary primate RPE cells, and human retinal explants.
Study takeaways
Retinal inflammation in AAV gene therapy is correlated with the vector dose. The inflammation is characterized by activation of antiviral innate immunity in the retina, which recruits a type 1 cell–mediated immune response. The use of a targeted immunomodulatory adjunct (in this case, hydroxychloroquine) during gene therapy may allow use of a lower vector dose, preventing development of retinal inflammation and improving clinical outcomes. •
References
The Association for Research in Vision and Ophthalmology. Understanding the mechanism of retinal inflammation following subretinal gene therapy. EventPilot Web. May 22, 2023. Accessed May 22, 2023. https://eppro02.ativ.me/web/page.php?nav=false&page=IntHtml&project=ARVO23&id=3878989
Xue K, Jolly JK, Barnard AR, et al. Beneficial effects on vision in patients undergoing retinal gene therapy for choroideremia. Nat Med. 2018;24(10):1507-1512. doi:10.1038/s41591-018-0185-5
Cehajic-Kapetanovic J, Xue K, Martinez-Fernandez de la Camara C, et al. Initial results from a first-in-human gene therapy trial on X-linked retinitis pigmentosa caused by mutations in RPGR. Nat Med. 2020;26(3):354-359. doi:10.1038/s41591-020-0763-1
Chandler LC, Barnard AR, Caddy SL, et al. Enhancement of adeno-associated virus-mediated gene therapy using hydroxychloroquine in murine and human tissues.Mol Ther Methods Clin Dev. 2019;14:77-89. doi:10.1016/j.omtm.2019.05.012
Chandler LC, McClements ME, Yusuf IH, Martinez-Fernandez de la Camara C, MacLaren RE, Xue K. Characterizing the cellular immune response to subretinal AAV gene therapy in the murine retina. Mol Ther Methods Clin Dev. 2021;22:52-65. doi:10.1016/j.omtm.2021.05.011
Kanmin Xue, MB-PhD, FRCOphth
e: kanmin.xue@eye.ox.ac.uk
Xue has no financial interest in this subject matter.

Newsletter
Keep your retina practice on the forefront—subscribe for expert analysis and emerging trends in retinal disease management.