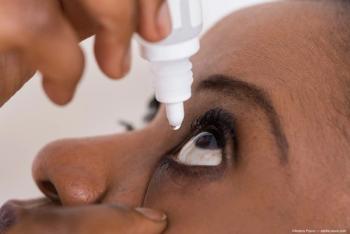
Eye drops developed by Columbia University researchers could prove to be a more effective and comfortable therapy for retinal vein occlusion (RVO).
New long-term data for Roche’s Vabysmo show sustained retinal drying and vision improvements in retinal vein occlusion

Eye drops developed by Columbia University researchers could prove to be a more effective and comfortable therapy for retinal vein occlusion (RVO).

If approved, RVO would be the third indication for faricimab-svoa in addition to wet age-related macular degeneration (AMD) and diabetic macular edema (DME).
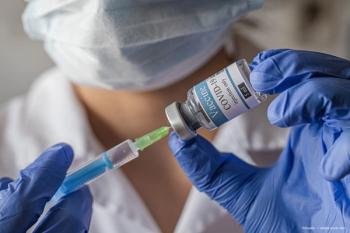
A large cohort study found no association between the first messenger RNA COVID-19 vaccinations and acute retinal vein occlusions.