
New research highlights iron dysregulation's role in dry AMD, suggesting transferrin as a promising treatment to slow disease progression.

New research highlights iron dysregulation's role in dry AMD, suggesting transferrin as a promising treatment to slow disease progression.

K8 is a member of a new class of inflammasome-inhibiting drugs called kamuvudines.

Gene therapy revolutionizes retinal disease treatment, offering long-lasting solutions and reducing the need for frequent injections in patients with nAMD.

Geographic atrophy management evolves with proactive imaging, personalized therapies, and real-world insights, enhancing patient care and treatment outcomes.

nAMD treatment options may offer positive outcomes to patients by reducing fluid fluctuations associated with poorer outcomes.
Guest lecturer explores the emerging frontiers in ROP therapy and the evolving ethical, clinical, and technological considerations.

Ophthalmology advances with AI diagnostics and pediatric trauma insights, merging technology and compassion to enhance patient care and outcomes.
Choroidal dysfunction plays a crucial role in retinal diseases, highlighting its potential as a therapeutic target and the need for innovative treatments.

Dr. Sharon Fekrat presented a retrospective analysis assessing how antiplatelet and anticoagulant therapy relates to hemorrhage characteristics and outcomes.
Sharon Fekrat discusses the complexities of antithrombotic use in wet AMD, highlighting the need for individualized treatment and future guideline development.

Remote monitoring of age-related macular degeneration enhances patient care, reduces treatment burden, and leverages AI for personalized management.

Prof Dr Rajvardhan Azad explains how unified guidelines, AI-driven tools, and cross-disciplinary collaboration could transform outcomes for premature infants.
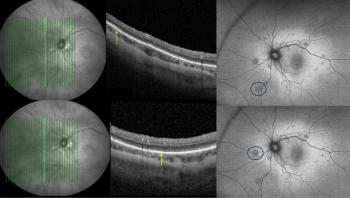
Priyanka Sanghi, BSc, MSc, MBBS, highlights how fundus autofluorescence and optical coherence tomography may help uncover long-term retinal footprints invisible to standard examination.

Innovative multimodal deep learning models enhance non-invasive chronic kidney disease screening by integrating retinal images and urine dipstick data for improved accuracy.

Dr. Melissa Yuan discusses the impact of weight and postmenstrual age on foveal maturation in preterm infants, highlighting critical insights for neonatal care.

The Portal extension trial reveals that the Port Delivery System significantly improves vision in AMD patients, showcasing long-term efficacy and durability.

New treatments show promise in preventing fibrosis in neovascular AMD, addressing a critical need for improved visual outcomes in patients.
Dr. J. Peter Campbell discusses the technologies shaping retinopathy of prematurity detection, clinical integration, and global implementation.

To help with education and awareness, Prevent Blindness is providing free, expert-approved educational resources on GA.

The company noted that this approval marks Celltrion's first Health Canada-approved biologic product in ophthalmology.

AAO 2025 highlights groundbreaking advancements in retinal disease treatments, showcasing senolytics, optogenetics, and personalized therapies for improved patient outcomes.
AAO 2025 revealed that true-color widefield imaging, AI-powered home OCT, and refined FAERS analyses are collectively transforming retinal diagnostics into a more precise, continuous, and safety-aware system.

According to the company, this is the first and only single-use kit on the market that includes the diagnostic lens.

Both therapeutics will leverage AGC Biologics’ BravoAAV suspension platform and use an innovative dual AAV vector approach, which splits the therapeutic gene into 2 halves.

EyePoint Pharmaceuticals' DURAVYU trials for wet AMD receive positive DSMC review, highlighting safety and potential for improved patient outcomes.

The two meetings will be held concurrently from December 4-7 in Florence, Italy.


For Jordan M. Graff, MD, FACS, early adoption is about reigniting the thrill of discovery and navigating the rewards, risks, and realities of translating innovation into practice.

