AMD
Latest News
CME Content


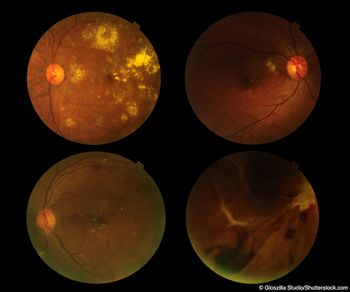

ICYMI: Abicipar Phase II MAPLE Trial supports improved safety for patients with nAMD following a Modified Manufacturing Process
David S. Boyer, MD, notes that the goal is to dry every patient out, with undertreatment the leading cause of a loss of visual acuity.


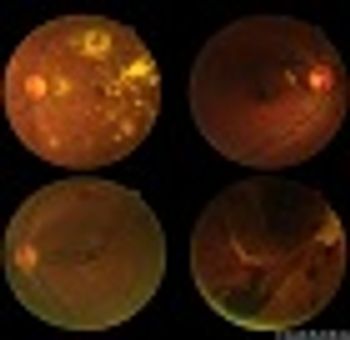




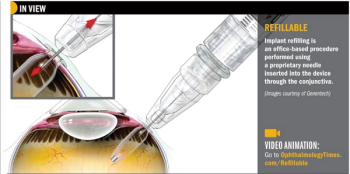
Investigators optimistic about sustained-release ranibizumab delivery based on phase II LADDER study

Patrik De Haes, MD, CEO of Oxurion NV, shares the latest updates in clinical studies at the OIS@AAO meeting in Chicago, Oct. 2018.



Although the phase III Chroma and Spectri studies for lampalizumab (Genentech), an investigational compound for the treatment of geographic atrophy secondary to age-related macular degeneration (AMD), failed to meet their primary endpoints, numerous lessons can be learned from the study data.
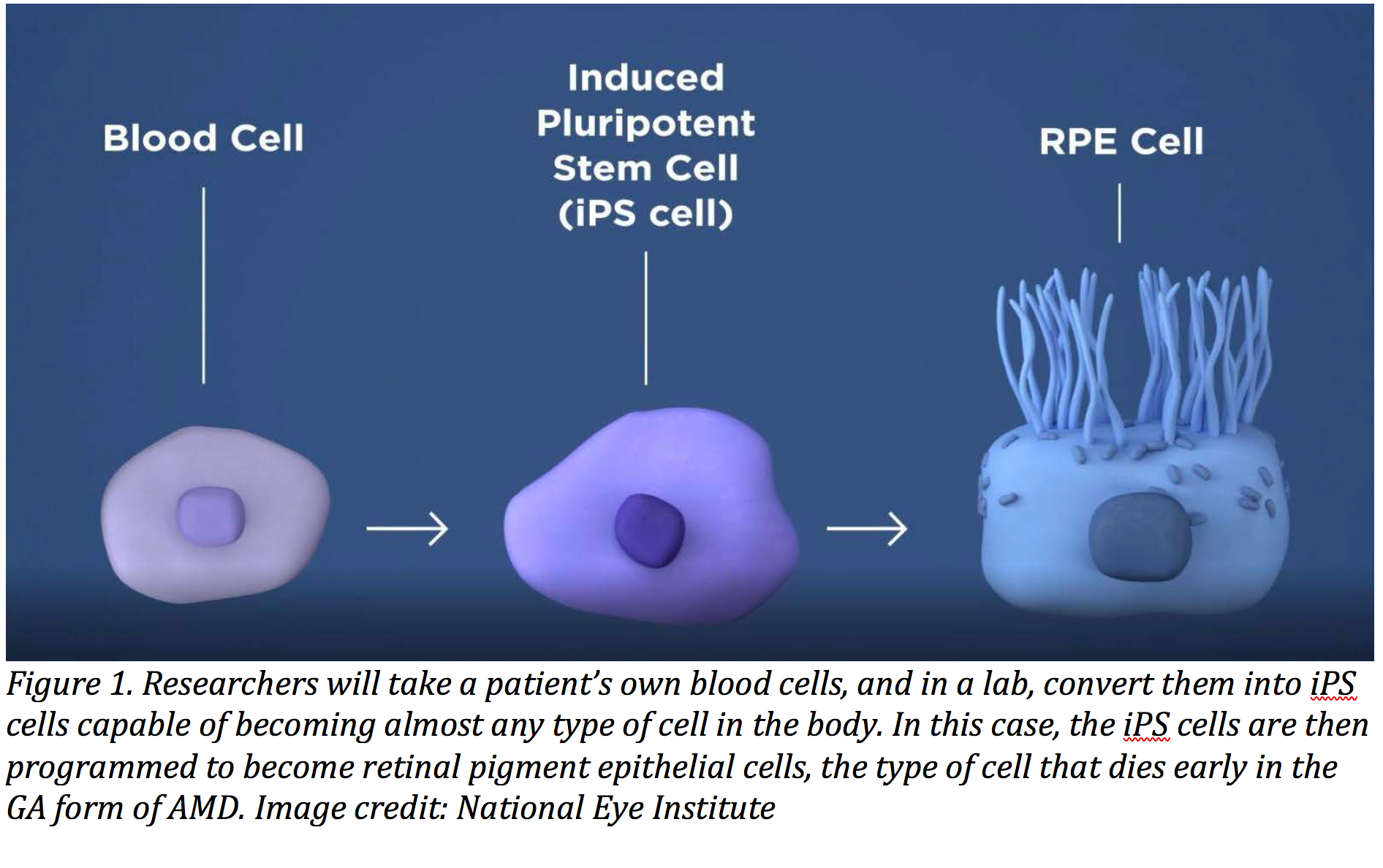
With the first ocular gene therapy approved in the United States, Szilard Kiss, MD, points out that the gene therapy era for retinal disease has arrived, “and hopefully it will arrive for age-related macular degeneration.”

Clinical trial data focusing on treatment for age-related macular degeneration (AMD) have been a highlight at many recent ophthalmology meetings. Here are a few of the latest clinical trials that physicians need to be aware.

Although a proven treatment therapy, age-related macular degeneration (AMD) requires frequent, costly anti-vascular endothelial growth factor (anti-VEGF) injections. Some pharmaceutical companies do have patient-assistance programs to help alleviate the financial burden.

A study has revealed that a budgetary saving per treated patient with the fluocinolone acetonide implant (Iluvien) versus ranibizumab (Lucentis) could lead to a significant cost-saving to NHS England.

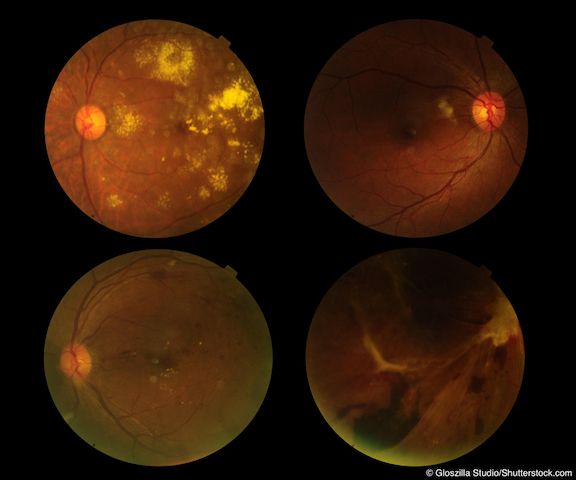
Twelve-month results of the FILLY trial show that in patients with geographic atrophy, the administration of complement C3 inhibitor APL-2 slowed the growth rate of the disease. It also appeared to increase the risk of new onset AMD, although this did not have an adverse effect on visual outcomes.

Research on treatments and/or causes of age-related macular degeneration (AMD) covers a wide range of approaches and paradigms. The latest research published in the past three months are perfect examples for understanding and treating AMD.