AMD
Latest News
Video Series

Latest Videos
Podcasts
CME Content
More News

The trial, sponsored by ONL Therapeutics, aims to enroll approximately 324 patients across sites in Europe, the United States, and Canada.
Early clinical experience highlights considerations for patient selection, surgical technique, and long-term monitoring in retinal disease.
Under the US agreement, OPUVIZ (aflibercept-yszy), a biosimilar to Eylea (aflibercept) 2 mg, may launch in January 2027.

Strategies for treatment intervals, drug selection, and patient expectations.

The Investigational New Drug (IND) application from Complement Therapeutics for CTx001 was previously approved by the FDA in October 2025.

A trio of retina specialists recently reviewed the clinical benefits of aflibercept 8 mg, including its extended dosing intervals, improved patient satisfaction, and enhanced treatment outcomes for various conditions.

Knowing what’s on the market for AMD and GA aids in preserving vision.
The novel system converts light into electrical signals to stimulate retinal cells.

The FDA noted in the letter that it is unable to approve the application for ONS-5010/LYTENAVA (bevacizumab-vikg) in its current form for the treatment of wet AMD.

Remote monitoring of age-related macular degeneration enhances patient care, reduces treatment burden, and leverages AI for personalized management.

New treatments show promise in preventing fibrosis in neovascular AMD, addressing a critical need for improved visual outcomes in patients.

Emerging anti-VEGF agents offer enhanced durability and anatomic outcomes in retinal disease.

New research highlights iron dysregulation's role in dry AMD, suggesting transferrin as a promising treatment to slow disease progression.

Xelafaslatide (formerly ONL1204) is a small-molecule Fas inhibitor designed to protect key retinal cells from cell death that occurs across multiple retinal diseases and conditions such as geographic atrophy.


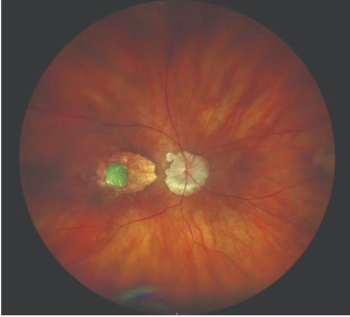
The investigators noted that this report is the first about subretinal drusenoid deposits in Black and Hispanic patients with age-related macular degeneration.

The newly approved technology delivers low-dose microcurrents through closed eyelids to stimulate retinal cells.

Luxa Biotechnology reveals promising phase 1/2a trial results for RPESC-RPE therapy, showing safety and potential vision restoration in patients with dry AMD.

Character Biosciences enhances its leadership team and secures $93 million in Series B funding to advance treatments for degenerative eye diseases.

Alteogen received a positive CHMP opinion for EYLUXVI in July 2025.

Belite Bio completes its phase 3 trial for tinlarebant, a potential first treatment for Stargardt disease, with results expected in late 2025.

Optain Health secures $26 million in Series A funding to enhance AI-driven retinal disease detection and expand its technology.
Researchers conducted this study to determine how often and why spontaneous soft drusen regresses without atrophy in patients who had intermediate or atrophic age-related macular degeneration.

Researchers will investigate a gene in the eye that is crucial for normal vision, but can cause retinal diseases when mutated that often lead to blindness.