
Study demonstrates results of pegcetacoplan as a geographic atrophy therapy.

Study demonstrates results of pegcetacoplan as a geographic atrophy therapy.
Although off-label repackaged bevacizumab syringes appear to satisfy an urgent clinical and financial need for patients with a variety of retinal disorders, they are known to be associated with considerable public health concerns due to the risks posed to patients from compounding pharmacies’ lack of compliance with a variety of FDA requirements.

After 10 years, AREDS2 formula shows increased efficacy compared to original formula, benefit of eliminating beta-carotene.
According to the study by a team of researchers from the University of California Irvine and University of Southern California, treatment with Humanin G reduced protein levels of inflammation markers that become elevated in age-related macular degeneration.

The study will investigate the safety, tolerability, pharmacokinetics, and efficacy of AM712 in subjects with neovascular age-related macular degeneration.

Biogen and Samsung Bioepis announce the launch of ranibizumab-nuna and detail when the ophthalmic biosimilar will be available for retina specialists.
The company noted that pegcetacoplan, designed to regulate excessive activation of the complement cascade, part of the body’s immune system, which can lead to the onset and progression of many serious diseases, was granted Fast Track designation by the FDA for the treatment of geographic atrophy.
Outlook Therapeutics initially filed the BLA in March or the use of bevacizumab-vikg in the treatment of wet AMD. The company said it will re-submit a revised BLA by September.

According to the company, the ReCLAIM-2 study of elamipretide demonstrates a correlation between ellipsoid zone dysfunction and vision.

A discovery by investigators at the National Eye Institute sheds light on tissue targeted by age-related macular degeneration and other diseases.

Professor Anat Loewenstein, MD, discusses data regarding the efficacy of faricimab at targeting both the VEGF and Ang2 pathways in patients with neovascular AMD and diabetic macular edema.

As more and more practices embrace dark adaptation testing, AdaptDx technology has become a staple in primary eye care.
Apellis revealed 18-month data for their investigational C3 therapy pegcetacoplan, unveiling continuous lesion growth reduction in eyes with geographic atrophy with monthly and every-other-month injections.
The data presented at ARVO showed single subcutaneous doses of D-4517.2 were safe, well-tolerated in healthy subjects.

According to the ALOFT study, patients demonstrate improved long-term vision in real-world setting after wet AMD conversion compared to current standard of care.

Jennifer I. Lim, MD, FARVO, FASRS, reviews the 2-year results of the YOSEMITE and RHINE trials, outlining the efficacy, durability, and safety of faricimab in diabetic macular edema.
The study found no association between treatment and the risk of chronic kidney disease or end-stage renal disease.
The axitinib intravitreal implant (OTX-TKI) is being evaluated to treat wet age-related macular degeneration in a phase 1b clinical trial.

A poll for retina specialists regarding their attendance at the 2022 Retina World Congress in Fort Lauderdale, Florida. This poll is now closed.

Apellis Pharmaceuticals releases results of their Geographic Atrophy Insights Survey (GAINS), revealing a large emotional toll and significant decrease in independence for patients with geographic atrophy.

A poll for retina specialists regarding their attendance at the Association for Research in Vision and Ophthalmology 2022 Meeting in Denver, Colorado. This poll is now closed.

Investigators found that patients with neovascular age-related macular degeneration in all countries included in the study lost vision as a result of the lockdown and reduced number of treatments during the COVID-19 pandemic.

If the Biologics License Application is approved by the FDA, the company could receive 12 years of marketing exclusivity for an FDA-approved alternative for the most frequently used anti-VEGF treatment in wet AMD patients in the United States.
The company notes that its clinical trial of the light delivery system meets the primary efficacy endpoint and can offer hope to patients with dry AMD who are experiencing vision loss and currently have limited treatment options.
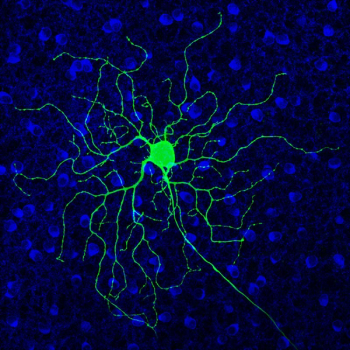
According to a team of investigators at University of California, Berkeley, tests of the drug Antabuse could prove the role of hyperactive retinal cells in blindness, potentially leading to better therapies.