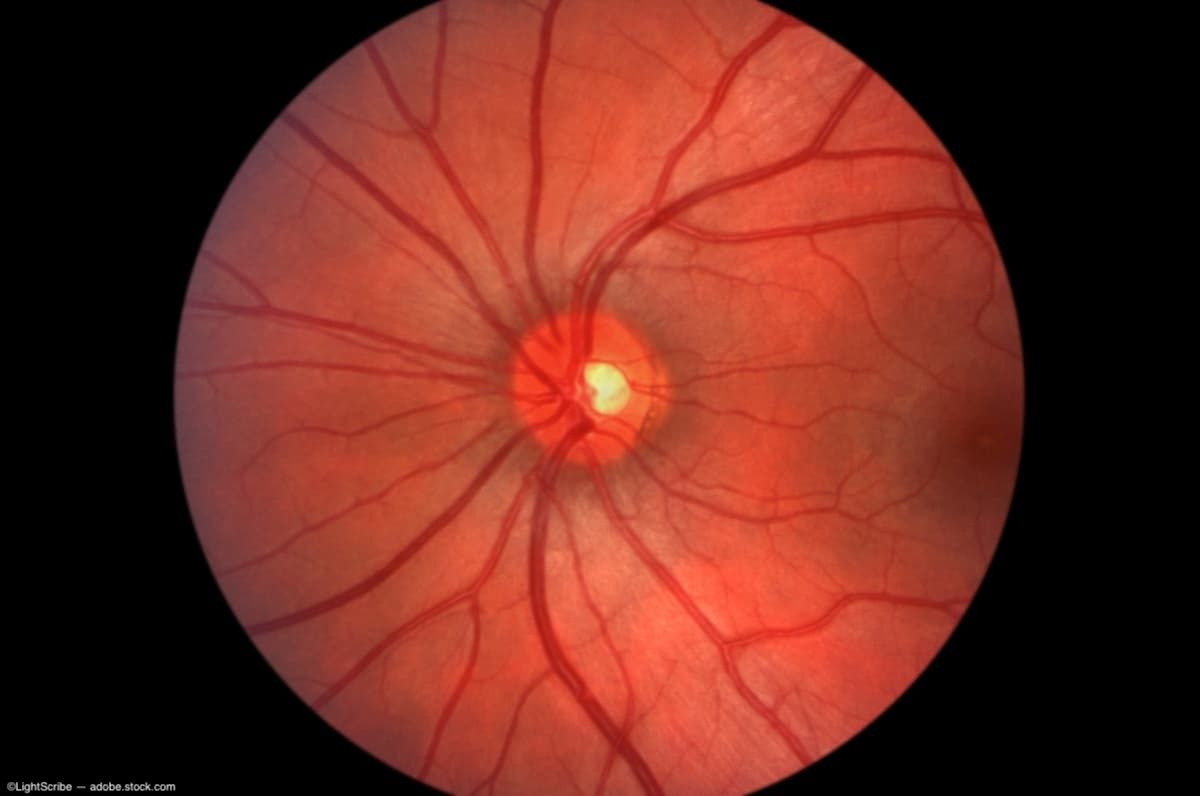
AMD
Latest News
Latest Videos

CME Content
More News
According to the company, its implantable miniature telescope is a Galilean intraocular telescope designed to improve visual acuity and quality of life for patients with late-stage AMD.

Deepak Sambhara, MD, shared an overview of his paper-on-demand, which covered real-world safety and efficacy of aflibercept, 8 mg in the treatment of neovascular age-related macular degeneration at the annual ASRS meeting in Stockholm, Sweden.

Yasha Modi, MD, spoke with Modern Retina about the ASRS 2024: The DAVIO phase 2 trial and upcoming LUGANO phase 3 trial at the annual ASRS meeting in Stockholm, Sweden.
Nathan Steinle, MD, spoke with Modern Retina about the ongoing research on the durability of sozinibercept in combination therapy with anti-VEGF-A treatments at the annual ASRS meeting in Stockholm, Sweden.

A new analysis by researchers at the National Institutes of Health shows the benefit of taking AREDS2 formula in late AMD.
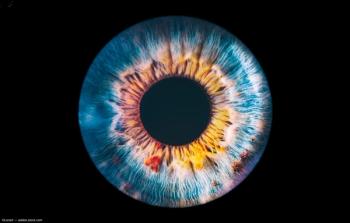
4D-150 Is being evaluated in wet age-related macular degeneration (AMD).

Adverum CEO Laurent Fischer spoke exclusively to Modern Retina about the 26-week interim analysis.
The key factor may be the control of inflammation.

Researchers from the UKB Eye Clinic, in cooperation with the University of Bonn and in close collaboration with basic and clinical scientists, have examined patients with early forms of AMD. The researchers focused on the so-called iRORA lesions, which are very early anatomical signs of retinal damage.

A novel formulation may prevent blood vessel growth and vascular leakage in the retina.
A team of researchers at the Ohio State University found that in mice an enzyme related to cell growth and division is a culprit in the blood vessel invasion in the back of the eye that causes blurred central vision in wet AMD.
The company announced it has received FDA approval to relaunch Susvimo for intravitreal use via an ocular implant for treating people with wet AMD.

The pharmaceutical candidate is being evaluated as a potential treat for nAMD.

The company expects approval from the EMA by 2025.
The biosimilar was approved for treating patients with age-related neovascular (wet) macular degeneration (nAMD) and other serious retinal diseases.

We asked what leaders in the retina field would tell residents and medical students as they train for the next decades in retinal care. Here's what 2 of them had to say.

According to the companies, the partnership will accelerate the development of lead product candidate, RO-104; a first-in-class tri-specific biologic for treating neovascular age-related macular degeneration.
A key distinction between patients seen in the clinic vs those enrolled in clinical trials has to do with characteristics dictated by the trial’s inclusion/exclusion criteria.

Researchers from the Case Western Reserve University School of Medicine and the Cole Eye Institute have found that supplement melatonin could be linked to a decreased risk of age-related macular degeneration.
Injection-free subgroup results demonstrated that a single intravitreal dose of 4D-150 without any supplemental anti-VEGF injections resulted in stable mean visual acuity that was equal to or higher than the standard bimonthly aflibercept control group at all 6 time points through Week 24.

The primary endpoint for the ReNEW and ReGAIN trials is the rate of change in the macular area of photoreceptor loss assessed by spectral domain-optical coherence tomography (SD-OTC) and ellipsoid zone mapping at week 48.

The IND approval will allow the company to initiate a Phase I/IIa clinical trial for its gene therapy treatment targeting wet Age-related Macular Degeneration (AMD) including Polypoidal Choroidal Vasculopathy (PCV).

According to the company, its Phase 3 program enrolled 1,984 patients across COAST and ShORe trials. Topline data from both pivotal trials are expected in 2025.
Investigators undertook a secondary analysis of the effects of long-term, low-dose aspirin in over 3,000 older adults who satisfied the inclusion criteria in the Aspirin in Reducing Events in the Elderly–AMD (ASPREE-AMD) study.

The agency approved Yesafili (Biocon Biologics) and Opuviz (Samsung Bioepis, Biogen) as biosimilars to Eylea (Regeneron Pharmaceuticals).